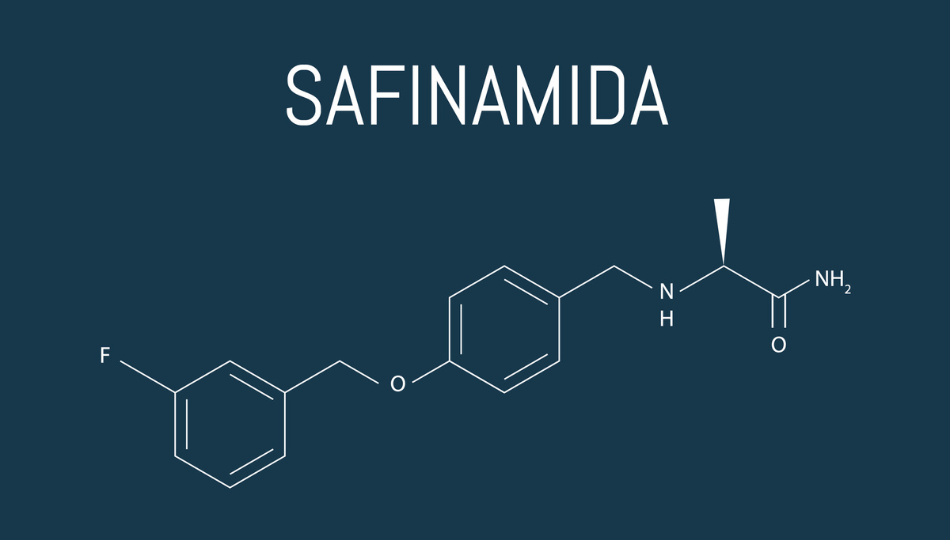
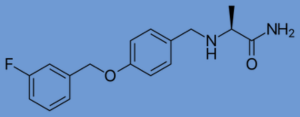
Safinamide is the newest MAO-B inhibitor approved for Parkinson’s. In 2017, the FDA approved safinamide for use in the US. It is approved for people with Parkinson’s who also take levodopa and experience troublesome OFF periods; it is not approved for use in isolation from levodopa.
Safinamide’s history began in the 1980s when researchers discovered a similar drug with anti-convulsant properties.
 What is an MAO Inhibitor, and How Does MAO Inhibition Help with Parkinson’s?
What is an MAO Inhibitor, and How Does MAO Inhibition Help with Parkinson’s?
Monoamine oxidase (MAO) is a family of enzymes involved in the metabolism of chemical messengers like dopamine and other neurotransmitters. They include MAO-A and MAO-B.
MAO-B breaks down dopamine, so inhibiting this enzyme helps dopamine last longer in your body.
Early MAO inhibitors were non-selective: They inhibited both MAO-A and MAO-B. This increased the risk of complications, primarily from increased organ stress and interactions with other drugs. Non-selective MAO inhibitors can cause high blood pressure when a person taking them consumes certain foods or certain medications.
Still, since MAO inhibitors have complex risk factors, please consult with your medical care team:
- About all medications you take before you start safinamide
- Before starting any new medication while taking safinamide
- Before stopping the use of safinamide
Safinamide’s Unique Characteristics as a Parkinson’s Treatment
Safinamide is an MAO-B inhibitor like rasagiline and selegiline, but has two important differences from these other drugs.
First, in addition to inhibiting MAO-B, safinamide also inhibits voltage-sensitive sodium channels and glutamate release. These effects may convey additional benefit in controlling Parkinson’s symptoms. For example, there is evidence that inhibiting glutamate release can help reduce dyskinesia.
Additionally, unlike rasagiline and selegiline, safinamide is reversible. This means it can detach from the MAO enzyme, improving its safety compared to rasagiline and selegiline–and these two drugs are widely considered safe and well-tolerated for most people.
![Figure 1. The novel mechanism of action from safinamide in Parkinson’s disease (PD) [14]. Sanchez Alonso, P.; De La Casa-Fages, B.; Alonso-Cánovas, A.; Martínez-Castrillo, J.C. Switching from Rasagiline to Safinamide as an Add-On Therapy Regimen in Patients with Levodopa: A Literature Review. Brain Sci. 2023, 13, 276. https://doi.org/10.3390/brainsci13020276](https://davisphinneyfoundation.org/wp-content/uploads/2023/09/Safinamide-2.png) Comparing Safinamide with Other Add-on Therapies
Comparing Safinamide with Other Add-on Therapies
A 2023 study found that switching from rasagiline to safinamide can reduce wearing-off.
A 2015 meta-analysis comparing safinamide and entacapone as add-on therapies reported that safinamide:
- Was well-tolerated with a safety profile not different from placebo, whereas comparison with placebo was significantly less favorable with entacapone
- Is typically taken once or twice daily; entacapone is commonly used between 3 and 10 times daily in clinical practice
- Has a lower incidence of side effects than COMT inhibitors
Although safinamide’s reversible quality lowers risk of side effects compared to selegiline and rasagiline, it remains active in the body longer than these other drugs. This may make its effects on symptoms more stable.
Moreover, while a 2020 study reported selegiline was the most effective MAO inhibitor as an add-on therapy, safinamide is also effective and avoids the risks associated with selegiline’s amphetamine metabolites.
Is Safinamide Neuroprotective?
Safinamide is not indicated for use as a neuroprotective or disease-modifying treatment. However, preclinical data from animal research suggests the drug has potential to convey a slight neuroprotective effect.
Two ways safinamide may convey neuroprotective effects are by suppressing microglial activation and by inhibiting glutamate. In response to inflammation, microglia have the capacity to damage neurons, and excess glutamate is associated with neurodegeneration.
Safinamide’s Risks
In clinical trials for safinamide, the most common side effects included dyskinesia, fall, nausea, and insomnia. Additional risks include:
High Blood Pressure
It is possible but unlikely that safinamide will increase blood pressure. The risk increases if you exceed the recommended dose because at higher doses, safinamide becomes less selective and can inhibit MAO-A and MAO-B. This causes dietary interactions–especially if you eat foods high in tyramine, like aged cheeses or meats.
Blood pressure elevation can also occur if you take safinamide with certain decongestants and cold remedies. Talk to your doctor before you start any new medications while taking safinamide.
 Serotonin Syndrome
Serotonin Syndrome
Safinamide slows serotonin metabolism, so when it is concurrently taken with other medications that have the same effect or increase serotonin levels, there is an elevated risk of serotonin syndrome.
This risk is generally low. In the clinical trials that led to safinamide’s FDA approval, only one case of serotonin syndrome was reported. Because serotonin syndrome can be deadly, caution should be used when concurrently taking serotonin reuptake inhibitors with safinamide.
Worsening Psychosis
The relationship between Parkinson’s and psychosis is complex and use of safinamide is not recommended for people with pre-existing psychosis.
Prenatal Risks and Risks to Nursing Children
There are no well-controlled studies of the use of safinamide by women during pregnancy. There are, however, some negative effects on offspring in animal studies. Accordingly, the prescribing information for the drug advises it should only be used during pregnancy if the potential benefit justifies the potential risk to the fetus.
Additionally, it is not known whether safinamide can be transmitted through breast milk but in animal studies, there were signs that the drug did pass through the mother’s milk. Use of the drug by nursing mothers is not advised.
Other Interactions and Risks
Visit the medications to avoid page of our medication guide for a list of medications known to have interactions with safinamide.
Cost
Safinamide is not available as a generic medication and the cost for a prescription is high. The manufacturer, Supernus, has a page on their website to help lower your cost, but before you attempt to fill a prescription, confirm your expected out-of-pocket costs with your insurance provider.
Reasons to Consider Safinamide
In addition to the reasons described above, other possible benefits of taking safinamide include:
Help with Depression and Apathy
A post-hoc analysis of a Japanese study of safinamide found it may improve depression and apathy during OFF times.
Decreasing Pain
A 2018 study found that safinamide was more effective than cannabinoids, dopamine agonists, COMT inhibitors, and Chinese medicine therapies at treating pain associated with Parkinson’s. The Japanese study mentioned above also had positive findings relative to the drug’s effect on pain. Further evidence for this effect arises from a 2017 study that appeared in the Journal of Parkinson’s Disease.
Possible Improvements for Dyskinesia
Because safinamide can extend the effect of levodopa, its use may increase dyskinesia. This is evident in the FDA’s prescribing information that reports dyskinesia as a common side-effect. However, authors of an analysis of one of the trials that led to approval of safinamide, found that “safinamide significantly increased daily ON time without increasing the risk of developing troublesome dyskinesia.”
Further, the authors share:
“For the overall treated population (with or without baseline dyskinesia), safinamide 100 mg/day significantly improved the dyskinesia rating scale score, compared with placebo, in the subgroup of patients with no change in levodopa dose. For patients with baseline dyskinesia, improvements over placebo were also significant in patients with or without changes in levodopa dose.”
Possible Improvements in Rapid Eye Movement Sleep Behavior Disorder (RBD)
A small 2021 study found that out of 30 participants who were experiencing RBD and took safinamide, 22 experienced “clear improvement” in their RBD symptoms. 16 participants were completely free from RBD symptoms.
Benefits to Motivation, Fatigue, and Urinary Symptoms
A small 2022 study involving 20 people taking 50mg of safinamide found broad benefit for non-motor symptoms, including regarding motivation, fatigue, and urinary symptoms associated with Parkinson’s.
Broad Positive Perspective from a Survey of 90 Movement Disorder Specialists
In 2022, NPJ published the results of a survey of 90 European movement disorder specialists about safinamide. The following list contains a sampling of statements with which over 80% of the specialists agreed. Safinamide:
- Reduces OFF time (100% agreed)
- Improves sleep/fatigue (80% agreed)
- Improves mood (83% agreed)
- Is effective for the management of pain (81% agreed)
- Improves quality of life (98% agreed)
- Is a valid therapeutic option in early stages of fluctuations (97% agreed)
- Is a valid therapeutic option for advanced Parkinson’s (98% agreed)
- Has a reversible effect which is considered advantageous (99% agreed)
Additional Resources
Medication Guide: COMT Inhibitors
WANT MORE PRACTICAL RESOURCES LIKE THIS?
You can learn much more about living well with Parkinson’s today through our Every Victory Counts® suite of resources. Each manual is packed with up-to-date information about everything Parkinson’s. Click the link below to order your manual(s).
Thank you to our 2023 Peak Partners, AbbVie, Amneal, and Kyowa Kirin, and our Every Victory Counts Gold Sponsor, AbbVie Grants, for their ongoing support of these must-have manuals. Additionally, we’d like to thank Barbara and Dale Ankenman, Abby and Ken Dawkins, Bonnie Gibbons, Gail Gitin in loving memory of Gene Gitin, Irwin Narter, and Lorraine and J Wilson for their generous donations that allow us to make these resources available and free to all.