In this video, Dr. Soania Mathur interviews Dr. Michael Okun to discuss the latest therapies for Parkinson’s, including RT-QUIC (Parkinson’s biomarker), subcutaneous dopamine pumps, vibrating gloves, rescue medications, DBS, and more. Below, you can find links to the video, a transcript, and an audio download, as well as a summary of the topics discussed in the conversation.
You can download a copy of the transcript here.
Note: This is not a flawless, word-for-word transcript, but it’s close.
You can download the audio for this webinar here: Emerging Therapies and Parkinson's Audio .
NOtes
Advancements in Diagnosis
Dr. Okun discussed the recent alpha-synuclein seed amplification assay biomarker study results. He shared information about the history of the seed assay method, and he shared some details about how proteins like alpha-synuclein can be isolated from biological samples to help in diagnosing conditions early, possibly even before symptoms are evident. Dr. Mathur observed that the seed assay method might influence the ability to more accurately define subtypes, and Dr. Okun added that the availability of this biomarker test might also result in changes to how stages of Parkinson’s are defined. More details are discussed in the video as well as in this blog post.
Treatments on the Horizon
Thinking about diagnosing a person before symptoms are evident prompted Dr. Mathur and Dr. Okun to discuss what is on the horizon relative to treatments that may prevent a person who is at high risk for developing Parkinson’s from actually developing Parkinson’s. Dr. Okun noted that this would be a disease modification effect, and he observed that so far, the trials that have shown signs of disease modification potential generally do so with regard to a specific outcome. For example, the amount of time between a diagnosis of Parkinson’s and the development of dyskinesia. He observed that the debate about rasagiline’s disease-modifying potential has not progressed much but that if rasagiline has a disease-modification effect, it is a small one. This is also the case with other treatments that have shown disease-modifying potential, like exenatide and ambroxol. This suggests that the best way forward may be to combine treatments that show small effects to produce a larger effect, as has been successful in treating cancer.
Subcutaneous Delivery of Dopamine Replacement
Dr. Mathur noted that improving quality of life is also an important outcome for some therapies. She brought up subcutaneous delivery of dopamine replacement as one emerging therapy that may have a significant impact on quality of life. Two new formulations of injectable carbidopa-levodopa, one from Neuroderm and one from AbbVie, are on the horizon. These treatments have shown promise in increasing ON time and may also decrease dyskinesias. One benefit of these therapies over the Duopa pump is that there would be no need for invasive surgery. One issue with the subcutaneous delivery of medications is that some people develop nodules under the skin at the injection site, and these can be irritating over the longer term.
Transdermal Treatment
Transdermal treatments-therapies that are absorbed through the skin–also have the potential to improve quality of life. Developing a transdermal system to deliver levodopa has proven difficult, but there is an FDA-approved dopamine agonist, rotigotine, which is delivered transdermally. Another medication that is available in a transdermal patch is rivastigmine, which can help with Parkinson’s dementia. One significant benefit of transdermal and subcutaneous treatments is that they can reduce the number of pills a person has to take.
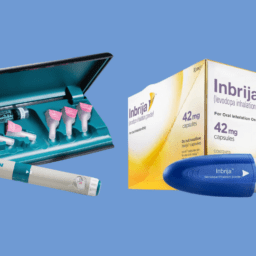
Rescue Medication
Dr. Mathur brought up apomorphine injections and rescue therapies to address OFF periods, and Dr. Okun observed that one benefit of treatments that are injected or inhaled, like inbrija, is that they can help clarify whether there a person’s microbiome may be influencing the efficacy of their medication. If your medication is not having a significant effect when taken in pill form but injected or inhaled rescue therapies work very well, your microbiome may be having an impact on drug absorption. He also noted that the important point is to try to not let an OFF period go on too long because a long OFF period can make it harder to return to ON.
Microbiome in Parkinson’s
While there is evidence that the microbiome is influential in Parkinson’s, the data is complicated, and there is no clear guidance as to how to address the microbiome’s influence on Parkinson’s. One complication is that the makeup of an individual’s microbiome changes very quickly and can even be different at different times of the day. Accordingly, Dr. Okun advised caution with trying to alter your microbiome as a therapy for Parkinson’s because there are many unknowns, many OTC probiotics are expensive, and doing so may make your Parkinson’s symptoms worse.
Inflammation
There is increasing information about the role of inflammation’s role in Parkinson’s. In particular, there is evidence that the use of certain prescription anti-inflammatory medications corresponds with a decreased incidence of Parkinson’s, but there is not enough information yet to recommend people with Parkinson’s use such medications for treatment.
Exercise
We continue to learn more about the positives of exercise for people with Parkinson’s. However, it is hard to isolate the degree to which exercise improves general health from the degree to which exercise influences Parkinson’s specifically. It is also complicated to establish the optimal dose and determine the best exercise for all people with Parkinson’s.
Still, we know that exercise is beneficial, and whether it is formally a disease-modifying treatment or whether it simply improves the quality of life is not especially significant. If, by exercising, you can maintain your lifestyle and avoid issues like falls or other health issues that may negatively impact your quality of life or complicate your experience of Parkinson’s, then this is significant and valuable.
Vibration Therapy
Vibration therapy was first observed to benefit Parkinson’s before there was even formal training in neurology. In the 1800s, Jean Martin Charcot observed that the vibrations experienced by his patients while on trains or carriages to visit with him seemed to produce a short-term alleviation of Parkinson’s symptoms. More recently, modern researchers have found that certain brain waves in people with Parkinson’s oscillate at unusual frequencies. This has led to research into whether we can manually adjust these waves by physically shaking the body. Prominent research on this subject has been undertaken at Stanford.
Evaluating DBS and other advanced therapy
There continue to be advancements in DBS and focused ultrasound therapy. There are now dual-lead and adaptive DBS systems, and there are studies looking at using focused ultrasound to produce lesions on both sides of the brain. There is some evidence that bilateral lesions produce a greater incidence of side effects, including side effects regarding speech and cognition. Focused ultrasound is less precise and tends to convey less significant symptomatic benefits.
Still, there is a role for focused ultrasound in some people, in part because the surgical risk is minimal compared to DBS. The important thing is that each person be evaluated by experienced clinicians to develop an individual treatment plan. The full picture of each individual’s experience of Parkinson’s should be considered before pursuing any therapy, especially an advanced one like DBS or focused ultrasound.
Sorting through Info
Dr. Okun observed that what is an emerging treatment will be different for each individual because each person’s experience will evolve over time. He also noted that there is so much information and so many emerging ideas about how to treat Parkinson’s–from chelation to vibration and supplementation. He encouraged everyone involved in a care team to be thoughtful in approaching new and emerging treatments, and he noted that there is an important role for independent foundations like the Davis Phinney Foundation to play insofar as they can serve as independent resources to help people think carefully about their care plan.
Additional Resources
The Importance of Finding Parkinson’s Biomarkers
Quaking our Way to a Parkinson’s Breakthrough at Parkinsonsecrets.com
Drug Repurposing for Parkinson’s: The Importance of Patience
About the SpeakerS
Dr. Soania Mathur
 Dr. Soania Mathur is a former family physician living outside of Toronto, Ontario, Canada, who resigned from her clinical practice twelve years following her diagnosis of young onset Parkinson’s at age 28. Now a dedicated speaker, writer, educator, and Parkinson’s advocate, Dr. Mathur is an active speaker in Canada and internationally at patient-directed conferences and has authored many published papers and online pieces that focus on patient education and empowerment. She serves as Co-Chair for the Patient Council of The Michael J. Fox Foundation for Parkinson’s Research, is a member of their Executive Science Advisory Board, is a member of the Board of Directors at The Davis Phinney Foundation, is part of the Editorial Board for the Journal of Parkinson’s Disease, and serves on The Brian Grant Foundation Advisory Board.
Dr. Soania Mathur is a former family physician living outside of Toronto, Ontario, Canada, who resigned from her clinical practice twelve years following her diagnosis of young onset Parkinson’s at age 28. Now a dedicated speaker, writer, educator, and Parkinson’s advocate, Dr. Mathur is an active speaker in Canada and internationally at patient-directed conferences and has authored many published papers and online pieces that focus on patient education and empowerment. She serves as Co-Chair for the Patient Council of The Michael J. Fox Foundation for Parkinson’s Research, is a member of their Executive Science Advisory Board, is a member of the Board of Directors at The Davis Phinney Foundation, is part of the Editorial Board for the Journal of Parkinson’s Disease, and serves on The Brian Grant Foundation Advisory Board.Dr. Michael Okun
 Dr. Michael Okun received his medical degree with honors from the University of Florida. He was fellowship-trained by Mahlon DeLong, Jerrold Vitek, and Ray Watts at Emory University in Atlanta, Georgia, before founding the movement disorders program at the University of Florida. He is currently Chair of Neurology, Professor, and Executive Director of the Norman Fixel Institute for Neurological Diseases at the University of Florida Health College of Medicine. He is a co-author of Ending Parkinson’s, Living with Parkinson’s Disease, and Parkinson’s Treatment: 10 Secrets to a Happier Life.
Dr. Michael Okun received his medical degree with honors from the University of Florida. He was fellowship-trained by Mahlon DeLong, Jerrold Vitek, and Ray Watts at Emory University in Atlanta, Georgia, before founding the movement disorders program at the University of Florida. He is currently Chair of Neurology, Professor, and Executive Director of the Norman Fixel Institute for Neurological Diseases at the University of Florida Health College of Medicine. He is a co-author of Ending Parkinson’s, Living with Parkinson’s Disease, and Parkinson’s Treatment: 10 Secrets to a Happier Life.Thank You to Our 2023 Live Well Today Webinar Series Presenting Sponsors
*While the generous support of our sponsors makes our educational programs available, their donations do not influence Davis Phinney Foundation content, perspective, or speaker selection.