Clinical trials are essential not just for finding a cure for Parkinson’s but for learning how to live well today. However, there are still a lot of misconceptions about clinical trials; so, we’d like to clear them up and share a few reasons why you might want to get involved.
Myth #1: Clinical trials already have all the people they need
Many people think that clinical trials already have plenty of members and that their own participation is not important. That’s not true. Nearly 85% of trials are delayed because of a lack of participants. Since clinical trials are the best way to advance Parkinson’s medications and treatments, the fact that so many trials face these delays goes to show that your individual participation is essential to moving the needle forward.
Myth #2: Clinical trials only recruit for people who have been newly diagnosed
Many people worry that trials only want people who have been newly diagnosed with Parkinson’s and are not already taking medication; however, the great variety of trials means there is a need for all sorts of volunteers, including people who are already taking Parkinson’s medication. For people who are nervous or hesitant about participating in medical research, Davis Phinney Foundation Ambassador Bart Narter says, “Many studies involve only one or two days and involve nothing riskier than a blood draw. Isn’t that worth it to help move research forward?”
Myth #3: All clinical trials involve taking unproven medicine
Some people discount clinical trials altogether because they think they all involve taking medication, but there are many trials that are focused on diet, exercise or bodily fluctuations, and trials that don’t involve medications at all. Depending on what you want to be involved in, there are a variety of options.
Myth #4: Clinical trials are too complicated
Clinical trials can be confusing because of the terminology that they use. However, once you learn the most frequently used terms, it will seem like a much less intimidating process:
- Allocation: a method used to assign participants to a group in a clinical study
- Randomized allocation: participants are assigned to their treatment group by chance
- Nonrandomized allocation: participants are assigned to their treatment group not by chance
- Baseline characteristics: data collected at the beginning of a clinical study for all participants, such as age, sex/gender, race, ethnicity and study-specific measures
- Clinical studies: research studies involving human participants that are intended to add to medical knowledge
- Interventional studies (clinical trials): participants are assigned to groups that receive one or more treatment, or no treatment, so that researchers can evaluate the effects of the interventions
- Observational studies: participants are identified as belonging to study groups and are assessed for outcomes, and the investigator does not assign participants to specific treatments
- Eligibility criteria: the requirements that participants must meet or the characteristics they must have, including inclusion criteria (required for a person to participate) and exclusion criteria (prevents a person from participating)
- Funder type: describes the organization that provides funding or support for a clinical study, including activities related to design, implementation, data analysis or reporting
- Informed consent: a process used by researchers to communicate the risks and potential benefits of participating in a clinical study to participants
- Intervention/treatment: a process that is the focus of a study, including drugs, medical devices, procedures, vaccines and other products or noninvasive approaches such as education, modifying diet and exercise
- Intervention model: the general design of the strategy for assigning interventions to participants in a clinical study
- Cross-over assignment: groups of participants receive two or more interventions in a specific order, so all participants “crossover” to the other treatment and receive both treatments in different orders depending on their group
- Factorial assignment: groups of participants receive one of several combinations of interventions, so all possible combinations of treatments are given to different groups of participants
- Parallel assignment: two or more groups of participants receive different interventions “in parallel” to participants in other groups
- Single group assignment: all participants receive the same intervention
- Masking: one or more parties involved in the trial, such as the investigator or participants, do not know which participants have been assigned which interventions
- Phase: the stage of a clinical trial studying a drug or biological product, based on definitions developed by the US Food and Drug Administration, based on the study’s objective, the number of participants and other characteristics. For a complete explanation of each of the phases, go here.
- Placebo: an inactive substance or treatment that looks the same as, and is given in the same way as, an active drug or intervention/treatment being studied
- Sham comparator arm: a group of participants receives a procedure or device that appears to be the same as the actual procedure or device being studied but does not contain active processes or components
Myth #5: Clinical trials are too risky
Trials tend to make people nervous because they don’t know what to expect. However, all clinical trials are required to include a protocol, which is the researchers’ plan for what will happen. Protocols include:
- Why the researchers are conducting the study
- The eligibility and number of participants
- The schedule they will follow
- The length of the study
- The information they will collect about participants
These protocols exist to safeguard the participants through the informed consent process, which you can read about above in the glossary. In addition, all studies must be approved by an Institutional Review Board (IRB) that evaluates the study to make sure that the rights and welfare of participants are protected and that the researchers are upholding the utmost in ethical conduct throughout the entire research process (from recruiting participants to analyzing and reporting the data).
Although all clinical trials are different, there are some consistencies across all trials. As described above, all trials are vetted by review boards to make sure that the study is ethical, risks are minimized, and participants’ safety and privacy are respected. In addition, as a participant in a clinical trial you always have the right to ask questions before, during and after the trial to make sure that you fully understand what is happening. You also have the right to withdraw from the study at any point, for any reason, and without any repercussions.
Many of our Foundation’s Ambassadors are involved in clinical trials because they recognize the importance of volunteering in order to find a cure for Parkinson’s. Ambassador Bart Narter has been involved in 30 trials because he finds that they give him a sense of purpose:
“Parkinson’s research studies are something that I can do uniquely well, since I have Parkinson’s. Participating in such studies makes me feel less like a victim and more like an active and energized person who is fighting Parkinson’s.”
– Bart Narter
Ambassador Joe O’Connor, who has participated in 20 clinical trials, believes that they are an essential part of living well:
“First off, you will get a Parkinson’s screening at each test; so, you get regular feedback on how your Parkinson’s is progressing. Second, you are helping to find the cure! Third, you may get little cures for yourself, as I have experienced a couple of times. Finally, living well equals living cool, and as a result of these trials I get exposed to and in many cases I get to work with very cool, state-of-the-art medicine.”
– Joe O’Connor
Another of our Ambassadors, Rich Wildau, believes that participating in clinical trials is a form of engaging with the community:
“Living well means being socially engaged in a variety of ways. I have found that this kind of engagement is as important as exercise in slowing your personal disease process and growth of symptoms, so you gain as much as you give.”
– Rich Wildau
Myth #6: Clinical trials only happen in major cities
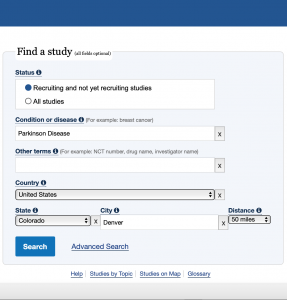
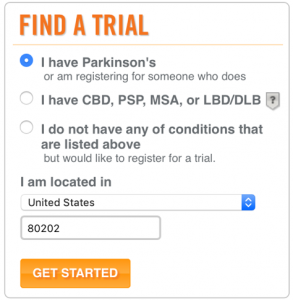
One other common misconception is that there aren’t any Parkinson’s clinical trials going on in your area. However, across the United States and throughout the world, there are thousands of clinical trials happening all the time. You can visit www.clinicaltrials.gov and fill out their “Find a study” box (as shown below) or visit foxtrialfinder.michaeljfox.org and fill out their “Find a trial” box (as shown below) and create an account to find clinical trials in your area.
Although our mission involves encouraging people with Parkinson’s to live well TODAY, we are also hopeful about and engaged in finding a cure for Parkinson’s. As such, we encourage people both with and without Parkinson’s to participate in clinical trials in the hopes that one day soon we will find a cure.
Further Resources
Michael J. Fox Foundation Clinical Trials Finder
Information about Clinical Trials and Suggested Questions for You to Ask
Full Glossary of Terminology for Clinical Trials
Want to learn more about the latest Parkinson’s research and clinical trials?
This October, we’re hosting a webinar titled The Latest Parkinson’s Research and Clinical Trials with Dr. Drew Kern. If you’d like to hear the most up-to-date information on these topics, register for free here.