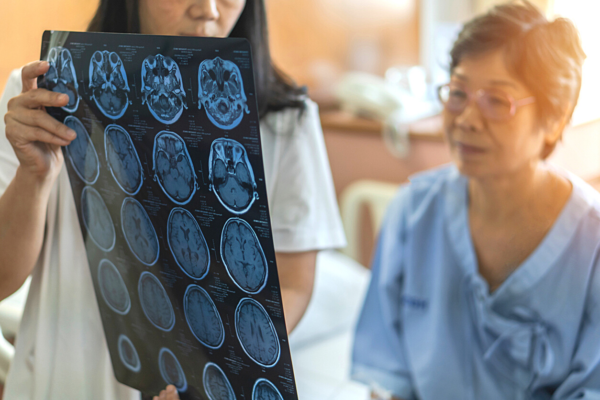
Focused ultrasound therapy (FUS) is an early-stage, single-procedure, non-invasive technology to manage tremor and dyskinesia. It is FDA-approved and is approved to treat tremor outside the US as well. It treats Parkinson’s symptoms through focused beams of ultrasonic energy that target specific areas deep in the brain, interrupting circuits involved with tremor and dyskinesia. Using a mechanism called ablation, the focused beams can disrupt targeted brain tissue to treat motor symptoms. Because focused ultrasound allows for precise targeting of areas of the brain, there is no damage to healthy tissue.
Other FUS mechanisms can temporarily disrupt the blood-brain barrier (BBB), allowing desired therapeutics access into the brain. This disruption of the BBB is also beneficial because it enables undesirable substances to leave the brain more easily.
Because FUS involves no incisions, holes in the skull, or electrodes in the brain, it can provide a powerful alternative to deep brain stimulation (DBS) with less risk of complications, as well as a lower cost. It does not require subsequent procedures, office visits for battery replacement, broken wire repair, or appointments to adjust simulator settings.
Researchers are currently exploring whether focused ultrasound can treat Parkinson’s underlying pathology and prevent progression and restore function. Preclinical studies suggest that FUS may restore function given its ability to temporarily open the BBB to improve the delivery of therapies such as anti-alpha synuclein antibodies, neuroprotective and neurorestorative drugs, and other therapies.
Recent focused ultrasound Research
Research is ongoing about FUS and its use as a Parkinson’s therapy. Read on for some recent news and findings about this treatment.
- After successful testing at the University of Virginia School of Medicine and other sites, the FDA has approved the use of focused ultrasound therapy for common Parkinson’s symptoms. This incisionless form of brain surgery, a less-invasive alternative to deep brain stimulation, has shown promise in treating mobility, rigidity, and dyskinesias in people with Parkinson’s.
- An FDA-approved incisionless thalamotomy procedure that uses focused ultrasound energy guided by magnetic resonance imaging (MRI) can help minimize tremor in people with Parkinson’s. The procedure, which typically takes two to three hours, relies on MRI to target the location in the brain responsible for the tremor; then an ultrasound helmet sends more than a thousand beams of energy through the skull to thermally ablate the area without damaging any nearby brain tissue.
- Novant Health has become the first health center in the South Atlantic US to offer MR-guided focused ultrasound, a non-invasive therapy that uses soundwaves to relieve tremor.
- An MR-guided focused ultrasound platform called Exablate 4000 (Exablate Neuro) has been approved in China to treat essential tremor and tremors caused by Parkinson’s. Because the technique requires no surgical incisions or anesthesia, it is expected to carry a lower risk than deep brain stimulation. The technology has received marketing approval from the US Food and Drug Administration (FDA), as well as from regulatory agencies in Argentina, Australia, Canada, Chile, Europe, Israel, Japan, Kazakhstan, Korea, Russia, Taiwan, and Turkey.
WANT MORE PRACTICAL ARTICLES LIKE THIS?
You can find much more in our Every Victory Counts® manual, packed with up-to-date information about everything Parkinson’s. Request your free copy here.
Thank you to our 2022 Peak Partners, Amneal, Sunovion, and Kyowa Kirin, as well as our Every Victory Counts Gold Sponsor AbbVie Grants, Silver Sponsor Lundbeck, and Bronze Sponsors Supernus and Theravance for helping us provide the Every Victory Counts manual to our community for free.